
The Future of Alzheimer’s
Treatment is CNS-Selective
Cnstyx (MSF) delivers targeted brain therapy without the peripheral toxicity that's
held back cholinergic treatment for 30 years. No $26K infusions. No GI devastation.
Just a daily oral pill.
The Current Treatment Landscape is Failing Families
Existing cholinesterase inhibitors (Aricept, Exelon, Razadyne) cause debilitating GI side effects because they hit peripheral acetylcholinesterase. Monoclonal antibodies cost $26,000/year and require IV infusion centers most families can't access.
6.9M
Americans living with
Alzheimer's
50%+
Discontinue current drugs
due to GI effects
$26K
Annual cost of Leqembi
infusions

CNS-Selective
Targets brain acetylcholinesterase while sparing peripheral systems. The selectivity ratio changes everything.
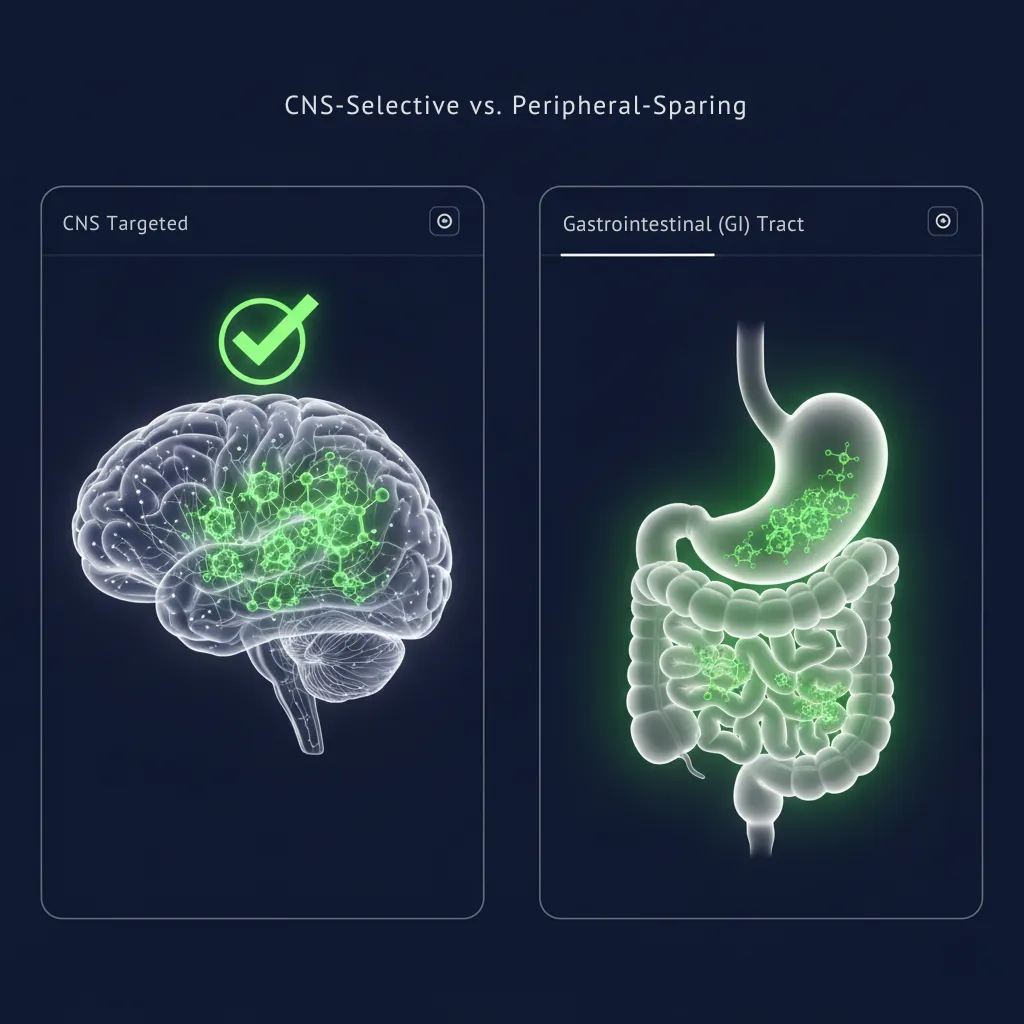
Peripheral-Sparing
No more nausea, vomiting, or diarrhea that forces patients off treatment. We removed the toxicity ceiling.

Oral & Accessible
A daily pill. No infusion centers. No $26K barrier. Treatment that can actually reach the 6.9 million who need it.
| Factor | Traditional AChE Inhibitors | Monoclonal Antibodies | Cnstyx (MSF) |
|---|---|---|---|
| CNS Selectivity | Low — hits peripheral systems | N/A — different mechanism | High — brain-targeted |
| GI Side Effects | Severe — 50%+ discontinuation | Moderate | Minimal — peripheral-sparing |
| Administration | Oral | IV Infusion required | Oral — daily pill |
| Annual Cost | ~$200–400 | ~$26,000 | Small molecule economics |
| Market Exclusivity | Generic — no protection | Patent protected | NCE Status — 5-year exclusivity |
| Adherence Barrier | High — side effects drive dropout | High — infusion access | Low — tolerable oral dosing |
"We've removed the toxicity ceiling that has held back Alzheimer's treatment for 30 years. CNS-selectivity isn't incremental improvement—it's the architectural change the field has been waiting for."
— Dr. Donald E. Moss, Founder & Chief Scientist
40+ years developing Cnstyx • Former UTEP neuroscience faculty

Dr. Donald E. Moss
Founder & Chief Scientist
40+ years developing MSF compound

John Norton
Clinical Trials Advisor
Former Dartmouth & Yale faculty

Hugo Sandoval
Asst. Chief Science Officer
Direct MSF research since 1990s

Phase I Data
Strong safety & tolerability data

NCE Status
5-year market exclusivity upon approval

Phase II Planning
Raising $5M for next stage